cGMP rHu IL-21 CSS
rHu IL-21 outfitted for closed-system manufacturing.
See why everyone is choosing Akron cytokines.
Akron’s Recombinant Human Interleukin-21 (rHu IL-21) is manufactured following all relevant cGMP guidelines for ancillary materials and is supported by a Type II Master File (MF), which can be referenced by the FDA during your drug or biologic application process. Akron’s rHu IL-21 is a single chain, 15.4 kDa, non-glycosylated cytokine analog expressed in E. coli, containing 132 amino acids. It is purified in a pharmaceutical facility without the use of histidine tags and nickel columns. Sterile filtration with aseptic filling and lyophilization are performed in-house with Endotoxin and Sterility testing performed per USP/EP on the final product; the lyophilized product is packaged in sterile vials.
IL-21 is expressed by activated helper T cells and NKT cells and is shown to enhance and sustain the populations and activity levels of NK cells and cytotoxic T cells. It is important for the proliferation and differentiation of T cells, B cells, and NK cells. The dimeric IL-21 receptor protein (IL-21R) shares an identical subunit with the IL-2, IL-7, and IL-15 receptor proteins and activates some of the same signal transduction mechanisms. IL-21 has significant structural homology with IL-2 and, like IL-2, has been used in the therapeutic treatment of cancer to suppress tumor growth. Akron’s cGMP-compliant rHu IL-21 can be used to promote the activation and proliferation of a range of cell types, including, but not limited to, CD4+ and CD8+ T cells, B cells, macrophages, monocytes, and dendritic cells (DCs).
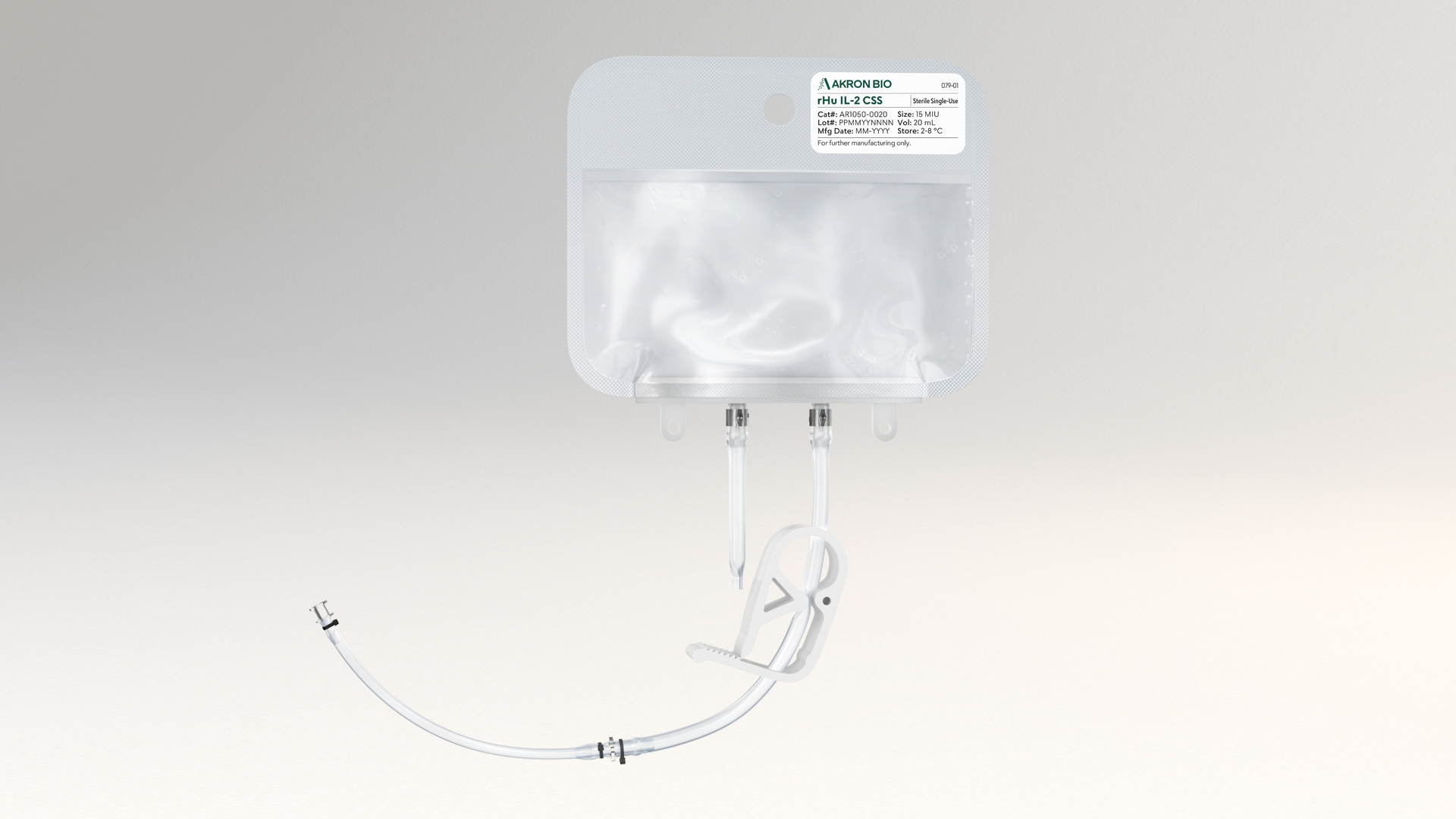
The liquid rHu IL-21 CSS product is packaged in a sterile fluoropolymer bag with two different weldable tubing connection options, allowing for easy incorporation into modern closed-system cell culture bioprocessing protocols. rHu IL-21 CSS increases safety and ease of use by eliminating the reconstitution step during manufacture and allows for the introduction of cytokine material into culture media in a fully contained manner to maximize sterility. Sterile filtration and aseptic filling are followed by Endotoxin and Sterility testing performed per USP/EP on the final product.
Active Substance
- Type II eCTD MF (#026085) on file with FDA
- Carrier protein-free formulation
- All raw materials are compliant, controlled, and traceable under Akron's Quality Management System (QMS)
Manufacturing
- Multi-step downstream purification strategy excluding affinity tags
- Commercial-scale production capacity
- E. coli expression system
- Sterile filtration and aseptic filling
Packaging
- Sterile bag chamber – 8-mil fluoropolymer film providing inert bio-compatibility, structural integrity, and minimal gas transmission
- Two-part tube weldable tube - proximal 6" portion made from weldable TPE AdvantaFlex® (1/8" ID x 1/4" OD), and the distal 6" portion made from standard weldable PVC (3/32" ID x 5/32" OD)
- All primary packaging is plasticizer free
- Primary packaging materials extensively validated, controlled, and qualified to ensure a consistent experience
- Every bag packed in individual protective cassette and shipped in validated CSafe Parcel insulated shipper
Quality
- Relevant cGMP guidelines used in manufacture, testing, and release
- USP <1043>, Ancillary Materials for Cell, Gene, and Tissue-Engineered Products
- EP 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products
- ISO 20399:2022, Biotechnology - Ancillary Materials Present During the Production of Cellular Therapeutic Products and Gene Therapy Products
Stability
- Under long-term stability program
- Store at 2-8 °C
- Transport with cold packs
- Do not freeze
Methods of Use
There are two different options available for connecting to and extracting the liquid solution from Akron's rHu IL-21 CSS product (for full instructions, see "Methods of Use" document):
- Weldable connection via proximal TPE AdvantaFlex® section (1/8" ID x 1/4" OD)
- Weldable connection via distal PVC section (3/32" ID x 5/32" OD)
For Use Statement
For research use or further manufacturing use in ex vivo cell therapy applications. This product is not intended for direct in vivo use or for direct clinical use as a drug, therapeutic, biologic, or medical device.
- Appearance
- pH
- ID via SDS-PAGE
- Biological Activity
- Purity via Reducing SDS-PAGE
- Purity via Non-reducing SDS-PAGE
- Bacterial Endotoxin
- Sterility
1. Why use Akron's Recombinant Human Interleukin-21 (rHu IL-21) Closed System Solutions (CSS)®?
Akron's rHu IL-21 CSS is a liquid cytokine formulation packaged in a sterile fluoropolymer bag with weldable tubing options for closed system processing. This format helps simplify manufacturing by eliminating the reconstitution step and allows the cytokine to be introduced into culture media in a fully contained manner. Akron's rHu IL-21 CSS is manufactured, tested, and released following relevant cGMP guidelines, and USP <1043> and ISO 20399:2022 have been considered in the manufacture of this product. The active substance is also supported by a Type II Master File on file with the FDA.
2. What is IL-21 and why is it used in cell therapy workflows?
Interleukin-21 (IL-21) is a cytokine primarily produced by activated CD4+ T cells and natural killer T (NKT) cells. It plays an important role in regulating immune responses and promotes the proliferation, differentiation, and functional activity of T cells, B cells, and natural killer (NK) cells. In cell therapy workflows, IL-21 is commonly used during ex vivo immune cell culture to enhance the expansion and functional activity of cytotoxic T cells and NK cells, often in combination with other cytokines depending on the specific manufacturing process.
3. What is the intended use of Akron's rHu IL-21 CSS?
For research or further manufacturing use in ex vivo cell therapy applications. This product is not intended for direct in vivo use or for direct clinical use as a drug, therapeutic, biologic, or medical device.
4. What organism is used to express Akron's rHu IL-21?
Akron's rHu IL-21 is produced using a genetically engineered E. coli strain.
5. How is Akron's rHu IL-21 different from native IL-21?
Akron's recombinant human IL-21 is a non-glycosylated cytokine expressed in E. coli. As a bacterially expressed recombinant protein, it lacks the glycosylation present in the native human protein and contains an N-terminal methionine. These differences are typical for cytokines produced using bacterial expression systems.
6. What excipients are used in Akron's rHu IL-21 CSS?
Akron's rHu IL-21 CSS is formulated in a citrate-based buffer containing sodium citrate dihydrate, mannitol, anhydrous citric acid, methionine, EDTA, and Tween 80 to support protein stability during storage and handling.
7. How is biological activity measured for Akron's rHu IL-21?
Biological activity is determined using a cell proliferation assay based on the Hybridoma B9 cell line. The ability of rHu IL-21 to stimulate proliferation of B9 cells is measured relative to a reference standard to estimate cytokine activity.
8. What release testing is performed on the final product?
Each lot of rHu IL-21 is tested against defined release specifications. The product specification includes appearance, pH, identity by SDS-PAGE, biological activity using a B9 hybridoma proliferation assay, purity by SDS-PAGE, bacterial endotoxins, and sterility. Typical specifications include appearance as a clear solution free of visible particles, pH within the defined specification range, purity of at least 90%, endotoxin not more than 250 EU/mg IL-21 per USP <85>, and sterility negative for growth per USP <71>.
9. What microbiological safety testing is performed?
Every lot of final product is tested for bacterial endotoxins using USP <85> and for sterility using USP <71>.
10. Do you have a Master File available for this product?
Yes. The active substance supporting Akron's rHu IL-21 CSS is backed by a Type II Master File on file with the FDA. A Letter of Authorization (LOA) may be available to support customer regulatory submissions.
11. Is there any BSE/TSE risk associated with Akron's rHu IL-21?
Because Akron's rHu IL-21 is expressed in E. coli, there is no BSE/TSE risk associated with mammalian expression systems. Akron provides TSE/BSE or Certificate of Origin documents upon request.
12. Are residual solvents used in the manufacture of Akron's rHu IL-21?
No. Residual solvents are not used in the manufacture of Akron's rHu IL-21.
13. What release and safety testing is performed on the final product?
Each lot is released against defined specifications that include appearance, pH, identity by SDS PAGE, identity by cell proliferation assay, purity by SDS PAGE, biological activity, bacterial endotoxins, and sterility. Endotoxin and sterility testing are performed using compendial methods.
14. Are recommended concentrations or dosing schedules available for this product?
No. Because each user's process is different, no recommended concentration or dosing schedule is provided. As described in USP <1043>, initial qualification of an ancillary material should include evaluation of dose-response effects on yield, purity, or potency of the therapeutic product to determine the optimal amount for the intended process.
15. What is the recommended storage condition for Akron's rHu IL-21 CSS?
Store at 2–8 °C. The product should not be frozen.
16. What are the shipping conditions for Akron's rHu IL-21 CSS?
This product ships with cold packs.
17. What is the shelf life for Akron's rHu IL-21 CSS?
The product has a 12-month interim shelf life supported by an ongoing long-term stability program. Stability testing is performed at 0, 3, 6, 12, 18, and 24 months, and the final target of the program is 24 months.
18. What packaging options are available for Akron's rHu IL-21 CSS?
The product is filled into a sterile fluoropolymer bag with two weldable tubing connection options. The top 6 inch portion is weldable TPE AdvantaFlex® (1/8 inch ID × 1/4 inch OD) and the bottom 6 inch portion is standard weldable PVC (3/32 inch ID × 5/32 inch OD).
19. What closed connection options are available for removing the material from the bag?
There are two options for making a weldable connection to the product, one through the proximal TPE section and one through the distal PVC section. The short sealed tube connected to the bag was used as the inlet tube and is not recommended for material removal.
IL-21 is expressed by activated helper T cells and NKT cells and is shown to enhance and sustain the populations and activity levels of NK cells and cytotoxic T cells. It is important for the proliferation and differentiation of T cells, B cells, and NK cells. The dimeric IL-21 receptor protein (IL-21R) shares an identical subunit with the IL-2, IL-7, and IL-15 receptor proteins and activates some of the same signal transduction mechanisms. IL-21 has significant structural homology with IL-2 and, like IL-2, has been used in the therapeutic treatment of cancer to suppress tumor growth. Akron’s cGMP-compliant rHu IL-21 can be used to promote the activation and proliferation of a range of cell types, including, but not limited to, CD4+ and CD8+ T cells, B cells, macrophages, monocytes, and dendritic cells (DCs).
Aliquot Sizes & Formats
Bags (100 μg) Cat. # AR1056-0100